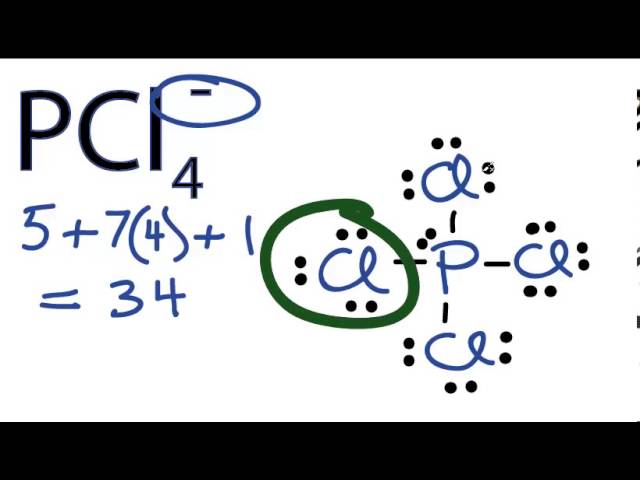
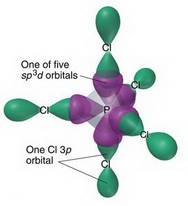
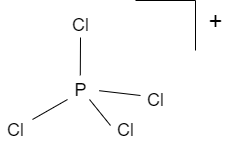
Consider the phosphorus tetrachloryl (PCl4+) cation. What is the central atom? Enter its chemical symbol. | Homework.Study.com

Predict the geometry and shape of Alcl4- , PCl4+ , Xef2 , ICL - Chemistry - Chemical Bonding and Molecular Structure - 16113665 | Meritnation.com

The shapes of PCl^+_4,, P{ Cl }^-_4 and AsCl_5 are respectively:tetrahedral, see-saw and trigonal bipyramidalsquare planar, tetrahedral and see-sawtetrahedral, square planar and pentagonal bipyramidaltrigonal bipyramidal, tetrahedral and square pyramidal
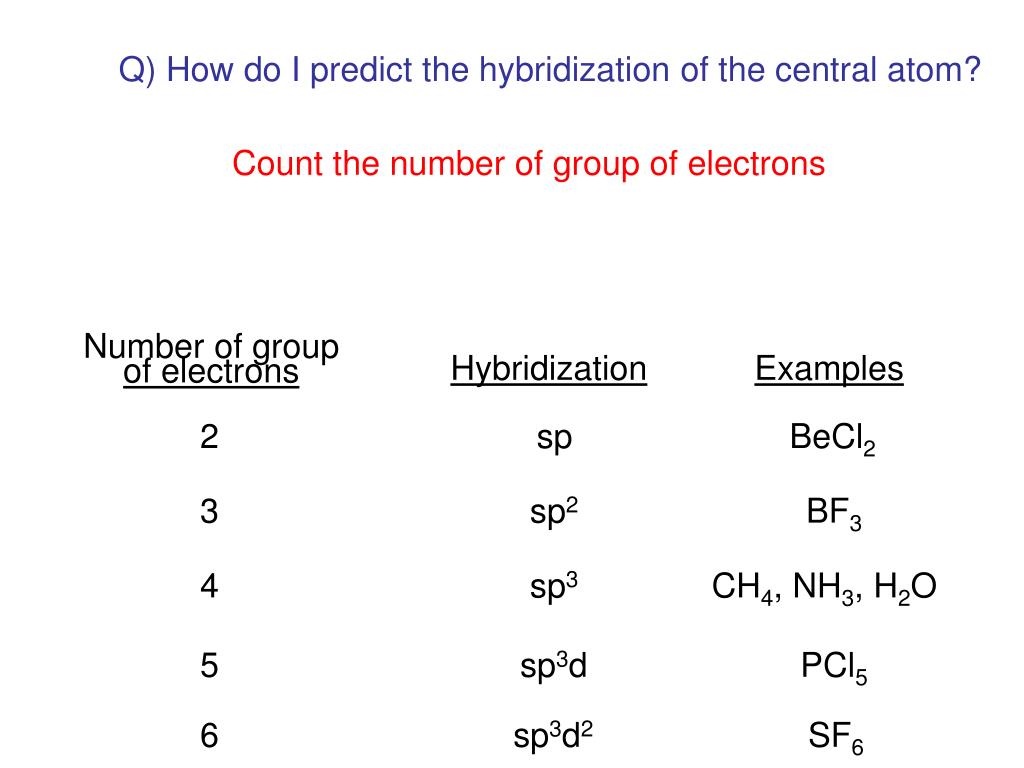
PPT - Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals PowerPoint Presentation - ID:5587869

Which of the following has a bond angle of approximately 120 deg? a) ClF3 b) SbBr6- c) PCl4- d) BeCl2 | Homework.Study.com
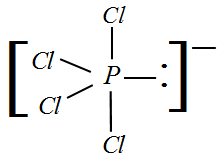
What are the shapes of $PC{l_4}^ + $ , $PC{l_4}^ - $ and $AsC{l_5}$ ?A. Square planar, tetrahedral and see-sawB. Tetrahedral, see-saw and trigonal bipyramidalC. Tetrahedral, square planar and pentagonal bipyramidalD. Trigonal

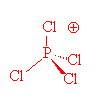
PCl4+ (Phosphorus tetrachloryl ion) Molecular Geometry, Bond Angles (and Electron Geometry) - YouTube

Orthophosphate and Sulfate Utilization for C–E (E = P, S) Bond Formation via Trichlorosilyl Phosphide and Sulfide Anions | Journal of the American Chemical Society

The shapes displaystyle:PCl_{4}^{+}, PCl_{4}^{-} and displaystyle:AsCl_{5} are respectively:square planar, tetrahedral and see-sawtetrahedral, see-saw and trigonal bipyramidaltetrahedral, square planar and pentagonal bipyramidaltrigonal bipyramidal ...
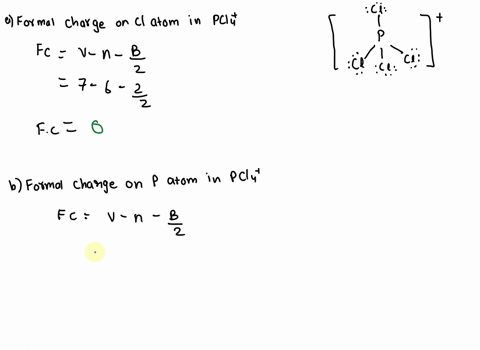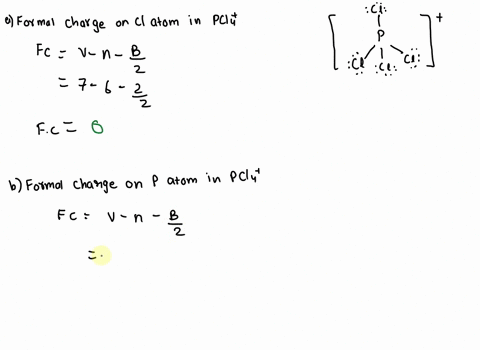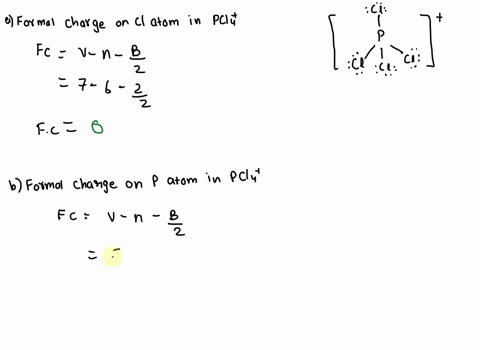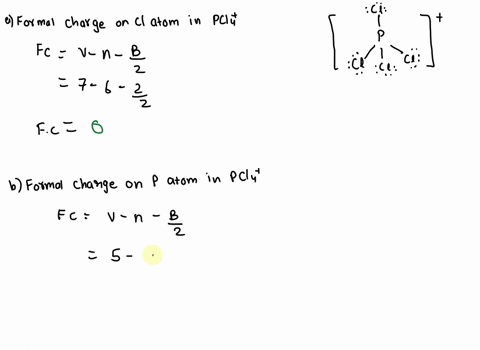
SOLVED: 2. What is the formal charge on the phosphorus (P) atom in PCl4+? 3. How do the P-Ci single bond lengths in PCl5, PCl4+, and PCl6- generally compare? 4. Identify the

![Kannada] Explain sp^(3)d hybridization PCl(5) molecule. Kannada] Explain sp^(3)d hybridization PCl(5) molecule.](https://d10lpgp6xz60nq.cloudfront.net/physics_images/SPH_BSR_CHE_QB_XI_C04_E03_022_S02.png)


_1.jpg)