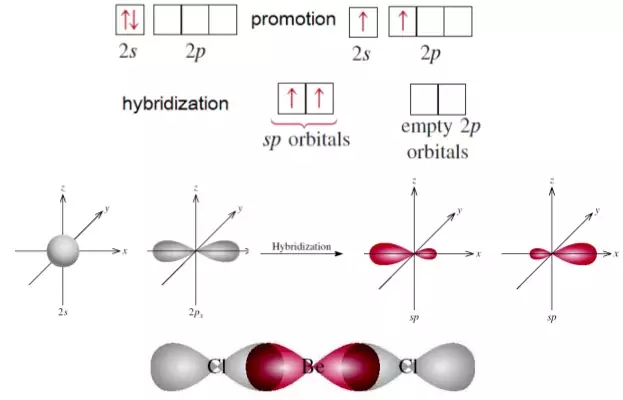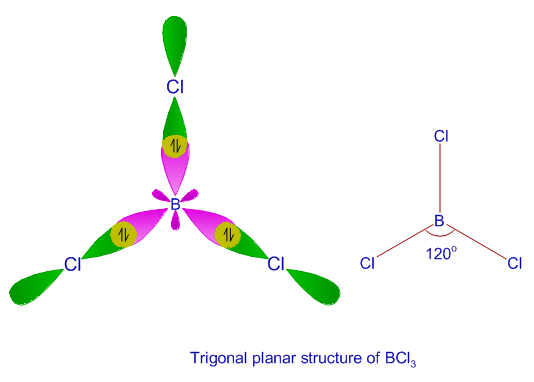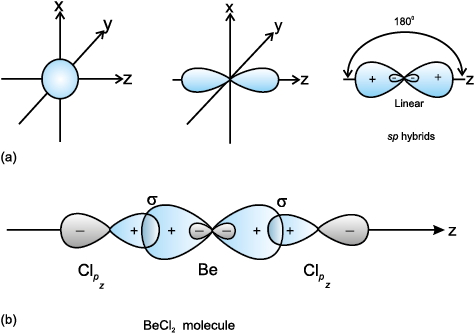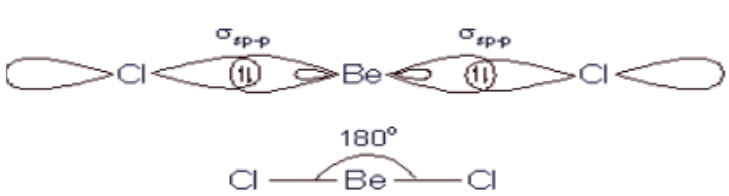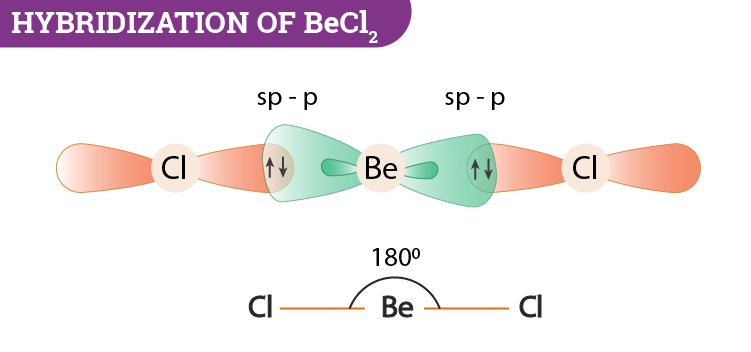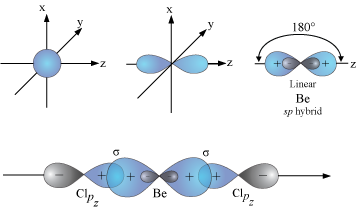
VSEPR theory tells that the valence electron pairs stay as far from each other as possible. For example, in BeCl_2 the two electron pairs of the two bonds are far apart so
47. What is the hybridization of BeCl2 in solid and vapour stage and give me the structures of both.

BeCl2 Lewis Structure, Molecular Geometry, Hybridization, Polarity, and MO Diagram - Techiescientist

Sir please explain this example of BeCl2 of sp hybridization - Chemistry - - 16269645 | Meritnation.com

BeCl2 Lewis structure, Molecular geometry, Hybridization, Bond angle and shape - Geometry of Molecules
Exemplars of Hybridization in Chemistry - askIITians Blog - One place for all updates on IIT JEE & Medical Exams
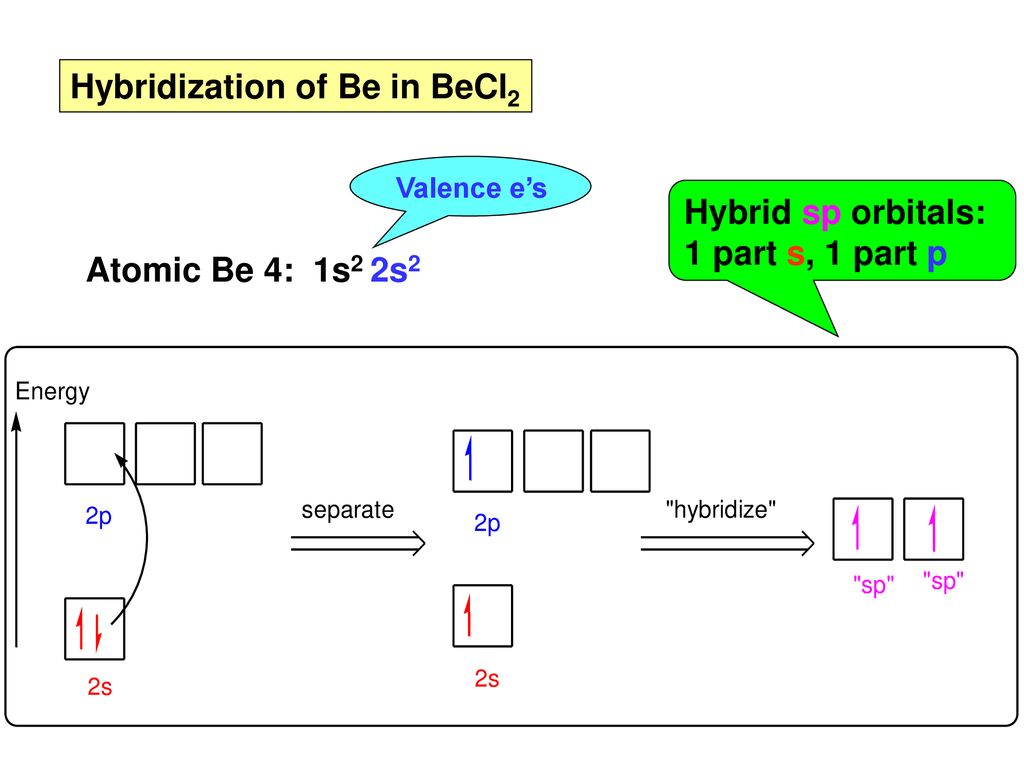
When does the Hybridization occur In becl2 there are 2 electron in 2s orbital of be then what - Chemistry - Electrochemistry - 13733947 | Meritnation.com