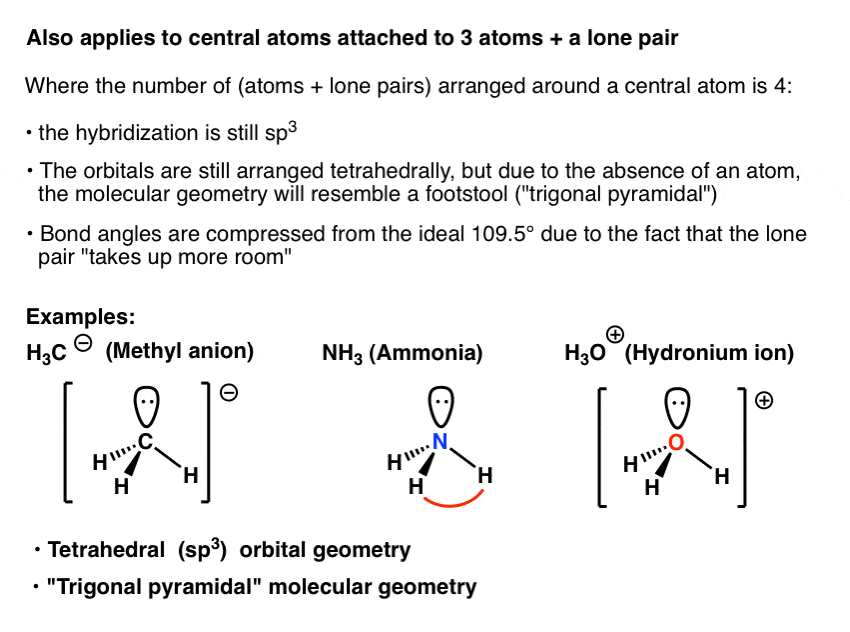
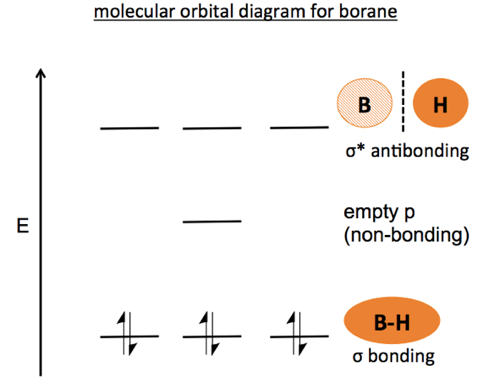
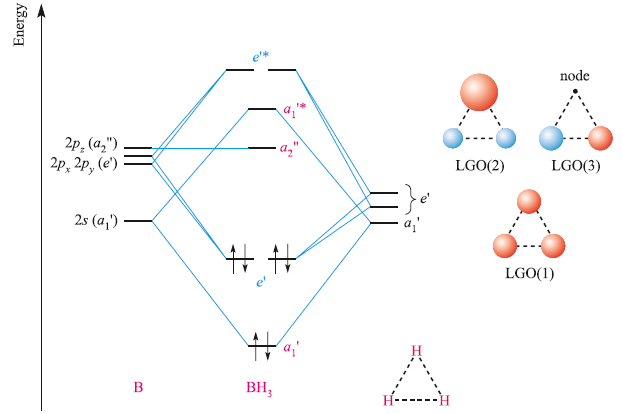
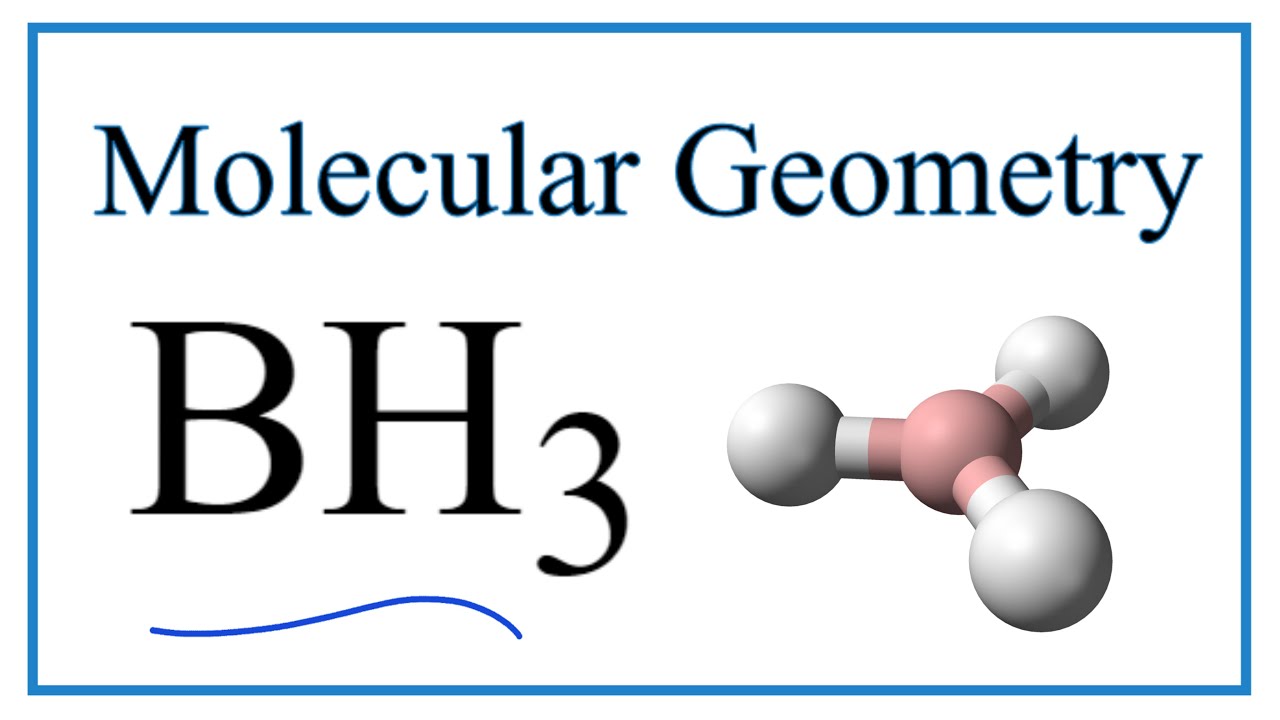
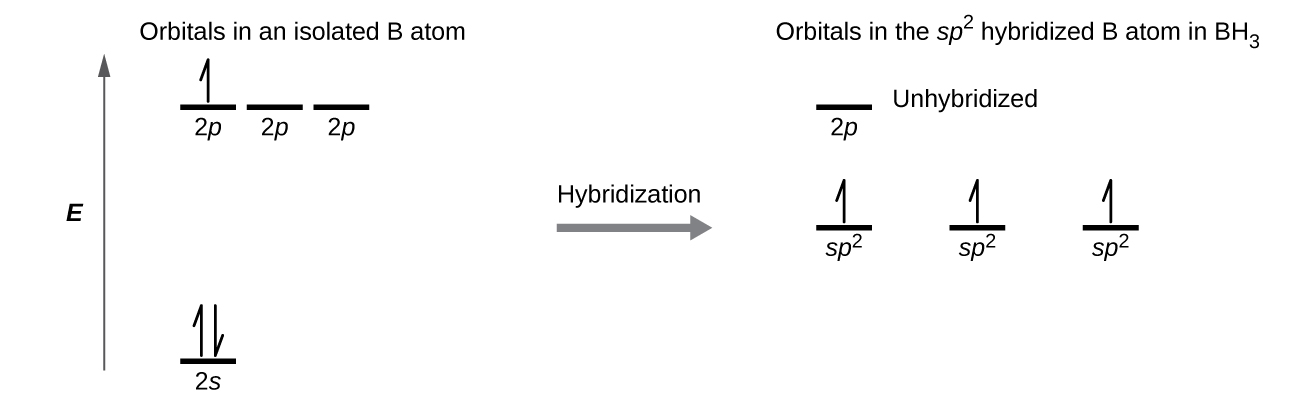
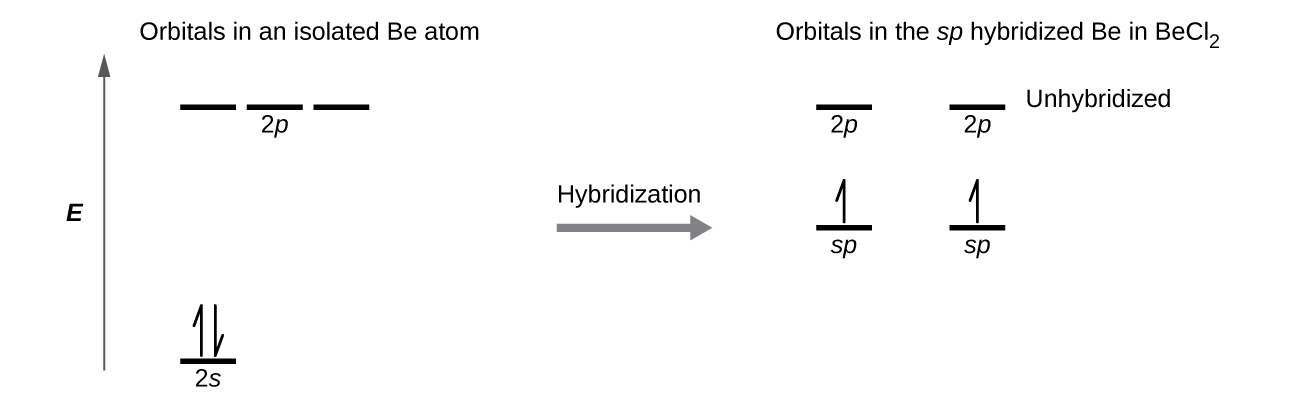
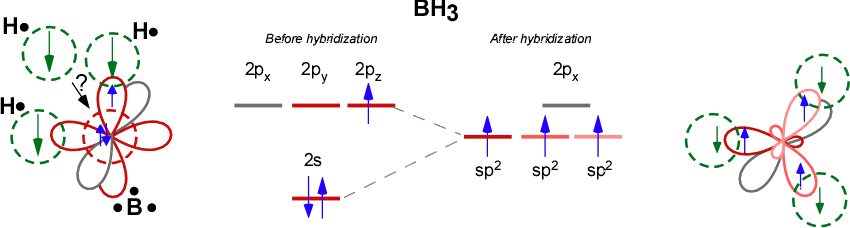
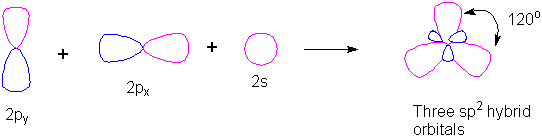SOLVED: (a) Using the lens pond theory, specify the type of hybridization on the central atom in the BH3 molecule. sp2-hybridization hybridization sp-d-hybridization (b) Draw orbital diagrams showing the distribution of orbitals,
Discuss the orbital structures of the following molecules on the basis of hybridisation, (i) BH3 (ii) C2H2 - Science City - Quora
Lewis dot structure and hybridisation of BH3 | Borane lewis structure and hybridization | JEE | NEET - YouTube
![The state of hybridization of the central atom in dimer of \\[B{H_3}\\] and \\[Be{H_2}\\] ?A. \\[s{p^2},s{p^2}\\]B. \\[s{p^3},s{p^2}\\]C. \\[s{p^3},s{p^3}\\]D. \\[s{p^2},sp\\] The state of hybridization of the central atom in dimer of \\[B{H_3}\\] and \\[Be{H_2}\\] ?A. \\[s{p^2},s{p^2}\\]B. \\[s{p^3},s{p^2}\\]C. \\[s{p^3},s{p^3}\\]D. \\[s{p^2},sp\\]](https://www.vedantu.com/question-sets/258693db-7817-497f-a15b-137735afe7095512141555770833478.png)
The state of hybridization of the central atom in dimer of \\[B{H_3}\\] and \\[Be{H_2}\\] ?A. \\[s{p^2},s{p^2}\\]B. \\[s{p^3},s{p^2}\\]C. \\[s{p^3},s{p^3}\\]D. \\[s{p^2},sp\\]

Discuss the orbital structures of the following molecules on the basis of hybridisation (i) BH3 (ii) C2H2 please answer fast - Chemistry - Chemical Bonding and Molecular Structure - 15719827 | Meritnation.com

The state of hybridisation of central atom in dimer of B{H}_{3} and Be{H}_{2}?s{ p }^{ 3 },s{ p }^{ 3 }s{ p }^{ 3 },s{ p }^{ 2 }s{ p }^{ 2 },